An HPLC system basically highly improved form of column chromatography. Instead of solvent being allowed to drip through a column under gravity, it is forced under high pressure of up to 400 atmospheres that makes the process faster. A typical HPLC consists of following components.
1. Pump
2. Injector
3. Column
4. Column oven
5. Detector
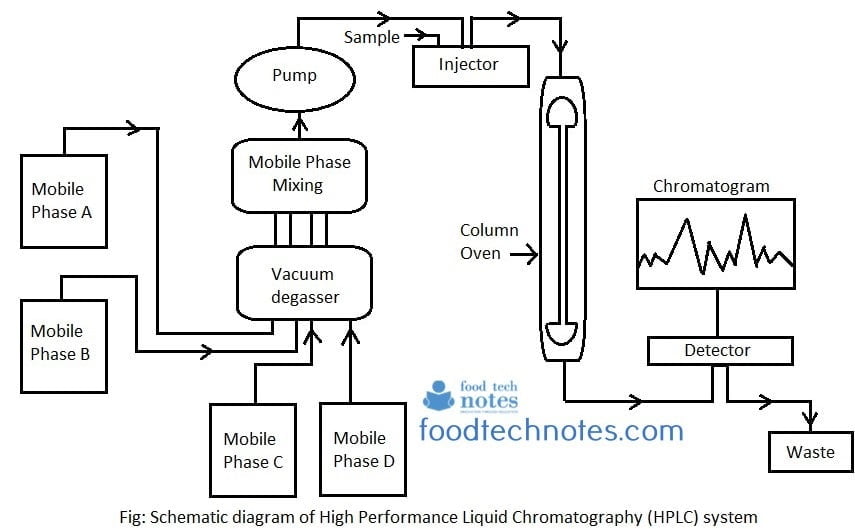
HPLC is a separation technique utilizing differences in distribution of compounds to two phases, called stationary phase and mobile phase. The stationary phase designates a thin layer created on the surface of fine particles and mobile phase designates liquid flowing over the particles. Under certain dynamic condition, each component in a sample has different distribution equilibrium depending on solubility in the phases and molecular size. As a result, components move at different speeds over the stationary phase and are thereby separated from each other.
The column is a stainless steel or resin tube which is packed with spherical solid particles. Mobile phase is constantly fed into column inlet at constant rate by liquid pump. A sample is injected from a sample injector, located near the column inlet. The injected sample enters the column with the mobile phase and the components in the sample migrate through it, passing between stationary and mobile phase. Components of sample move inside column only when is in mobile phase and move at different rate depending on
1. Molecular size / weight
2. Affinity to stationary phase / interaction to stationary phase
3. Solubility in mobile phase
Compounds that tend to be distributed in the mobile phase therefore migrate faster through the column while compounds that tend to be distributed in the stationary phase migrate slower. In this way, each component is separated on the column and constantly elutes from outlet. Each compound eluting from the column is detected by a detector connected to the outlet of column.
When separation process is monitored by the recorder starting at the time the sample is injected, a graph is obtained. This graph is called chromatogram. The time required for compound to elute is called retention time. The relationship between compound concentration and peak area depend on the characteristics of compound. Retention time is therefore used as an index for qualitative determination and peak surface area (height) as an index for quantitative determination.
About Author
Name : Pratiksha Shrestha
pratiksha.shrestha2001@gmail.com
Ms. Shrestha holds masters degree in food engineering and bioprocess technology from Asian Institute of Technology (AIT) Thailand. She is currently working for Government of Nepal at Department of Food Technology and Quality Control (DFTQC), Kathmandu. She is also a teaching faculty in College of Applied food and Dairy Technology (CAFODAT) affiliated to Purbanchal university, Nepal.
