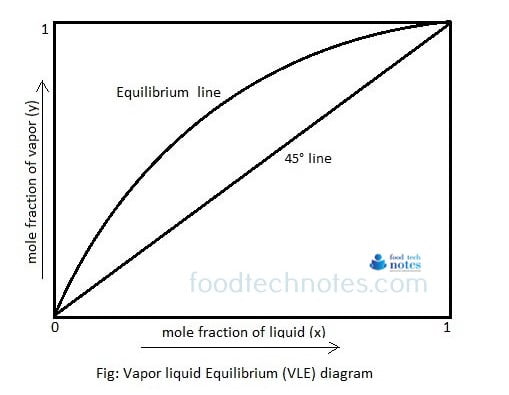
How mixtures can be separated on basis of relative volatility?
When a liquid mixtures is heated in a closed container, the mixture boils and starts forming vapor. The vapor will have different composition compared to the liquid. The vapors are obviously richer in more volatile component. If such vapors are collected and condensed, the condensed liquid will have different composition form original mixture and is richer in high volatile component. Read more